 Featuring eXtreme croquet and eXtreme croquet mallets.
Featuring eXtreme croquet and eXtreme croquet mallets.

 Dedicated to enjoying eXtreme croquet, nature, and the near-death experience!™
Dedicated to enjoying eXtreme croquet, nature, and the near-death experience!™






















Legal


|
 |

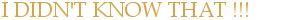
The Uncertainty Principle

By Sam Tudor
Essex, UK
The uncertainty principle, by Heisenberg, lies at the heart of quantum physics and sets it apart from the classical theories. The idea is that it is impossible to measure the position and the momentum of a particle precisely. The uncertainty in each of these areas, when multiplied together, must be greater than or equal to Planck's constant. Thus the more accurately you measure the position, the less exactly you can know its momentum. Particles do not have a definite position AND momentum.
Perhaps the same is true for croquet balls. Sometimes on long hit ins, the ball can appear to just hit another ball, but the stationary ball does not move. It is possible that we are concentrating on the momentum of the ball too much, thus allowing some uncertainty in the ball's position. For the stationary ball, we know for certain that its momentum is zero, allowing an uncertainty in its position equal to or greater than Planck's constant. The balls are not where they appear to be!
Copyright © by Sam Tudor. All rights reserved.
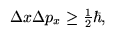
Heisenberg's Uncertainty Principle
WebMaster 
Home |
Intro |
Playing |
Rules |
Gallery |
Articles |
Mallets |
News |
Contact |
Links
Copyright © 2000 - 2020 by
Connecticut eXtreme Croquet Society
All rights reserved
|

|
|
|

